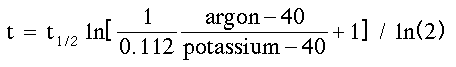Radiometric Dating
A Christian Perspective
Dr. Roger C. Wiens
Working at the Division of
Geological & Planetary Sciences
California Institute of
Technology
Presently working at the Space and Atmospheric Sciences
Group
Los Alamos National Laboratory
Home Address: 941 Estates Drive, Los
Alamos, NM 87544
rwiens@prodigy.net
Radiometric dating--the process of determining the age of
rocks from the decay of their radioactive elements--has been in widespread use
for over half a century. There are over forty such techniques, each using a
different radioactive element or a different way of measuring them. It has
become increasingly clear that these radiometric dating techniques agree with
each other and as a whole, present a coherent picture in which the earth was
created a very long time ago. Many Christians are completely unaware of the
great number of laboratory measurements that have shown these methods to be
consistent, and they are also unaware that Bible-believing Christians are
among those actively involved in radiometric dating. This paper describes in
relatively simple terms how some dating techniques work, how accurately the
half-lives of the radioactive elements and the rock dates themselves are
known, and how dates are checked with one another. In the process the paper
refutes some misconceptions prevalent among Christians today. God has called
us to be "wise as serpents" even in this scientific age. This paper is put out
by the American Scientific Affiliation and the Affiliation of Christian
Geologists to promote greater understanding and wisdom on this issue within
the Christian community.
TABLE OF CONTENTS
Introduction
Overview
The Radiometric
Clocks
Examples of Dating
Methods for Igneous Rocks
Potassium-Argon
Argon-Argon
Rubidium-Strontium
Samarium-Neodymium,
Lutetium-Hafnium, and Rhenium-Osmium
Uranium-Lead
Cosmogenic
Radionuclides: Carbon-14, Beryllium-10, Chlorine-369
The Age of the
Earth
Can
We Really Believe the Dating Systems?
Doubters Still
Try
Rightly
Handling the Word of Truth
Appendix: Common
Misconceptions Regarding Radiometric Dating Techniques
Further
Reading
Glossary
Introduction
Arguments over the age of the earth have sometimes been divisive in the
church. Although the Bible never mentions the earth's age, it is an issue
because some people have tried to calculate the date of creation by adding up
the life-spans of the generations listed in Genesis chapters 5 and 11. Assuming
a strictly literal interpretation of the week of creation, even if some
generations were left out of the genealogies, the earth would be less than ten
thousand years old. Radiometric dating techniques indicate that the earth is
thousands of times older than that--approximately four and a half billion years
old. Many Christians accept this and interpret the Genesis account in less
scientifically literal ways. However, some Christians suggest that the geologic
dating techniques are unreliable, that they are wrongly interpreted, or that
they are confusing at best. Unfortunately, there has not been much literature
easily available to Christians in an understandable form, so that confusion over
dating techniques continues.
The next few pages give a broad overview of
radiometric dating techniques, talk through a few examples, and discuss the
degree to which the various dating systems agree with each other. The goal is to
promote greater understanding in the Christian community on this issue. Many
people have been led to be skeptical of dating without knowing much about it.
For example, most people don't realize that carbon dating is not used on rocks
at all. God has called us to be "wise as serpents" (Matt. 10:16) even in this
scientific age. In spite of this, differences still occur within the body of
Christ. A disagreement over the age of the earth is relatively minor in the
whole scope of Christianity; it is more important to agree on the Rock of Ages
than on the age of rocks. But because God has also called us to wisdom, this
issue is worthy of study.
Overview

Rocks are made up of many individual
crystals, and each crystal is usually made up of at least several different
chemical elements such as iron, magnesium, silicon, etc. Most elements in nature
are stable and do not change. However, some elements are not completely stable
in their natural state. Some atoms eventually change from one element to another
by a process called radioactive decay. If there are many atoms of the original
element, called the parent element, the atoms decay to another element, called
the daughter element, at a predictable rate. The passage of time can be charted
by the reduction in the number of parent atoms, and the increase in the number
of daughter atoms.
Radiometric dating can be compared to an hourglass.
When the glass is turned over, sand runs from the top to the bottom. You cannot
predict exactly when any one particular grain will get to the bottom, but you
can predict from one time to the next how long the whole pile of sand takes to
fall. Once all of the sand has fallen out of the top, the hourglass will no
longer keep time unless it is turned over again. Similarly, when all the atoms
of the radioactive element are gone, the rock will no longer keep time (unless
it receives a new batch of radioactive atoms).
Unlike the hourglass,
where the amount of sand falling is constant right up until the end, the number
of decays from a fixed number of radioactive atoms decreases as there are fewer
atoms left to decay (see Figure 1). If it takes a certain length of time for
half of the atoms to decay, it will take the same amount of time for half of the
remaining atoms, or one-fourth of the original total, to decay. In the next
interval, with only one-fourth remaining, only one eighth of the original total
will decay. By the time ten of these intervals, or half-lives, have passed, less
than one thousandth of the original number of radioactive atoms is left. Also
unlike the hourglass, there is no way to change the rate at which radioactive
atoms decay on earth. If you shake the hourglass, twirl it, or put it in a
rapidly accelerating vehicle, the time it takes the sand to fall will change.
But the radioactive atoms used in dating techniques have been subjected to heat,
cold, pressure, vacuum, acceleration, and strong chemical reactions without any
significant change in their decay rate.1
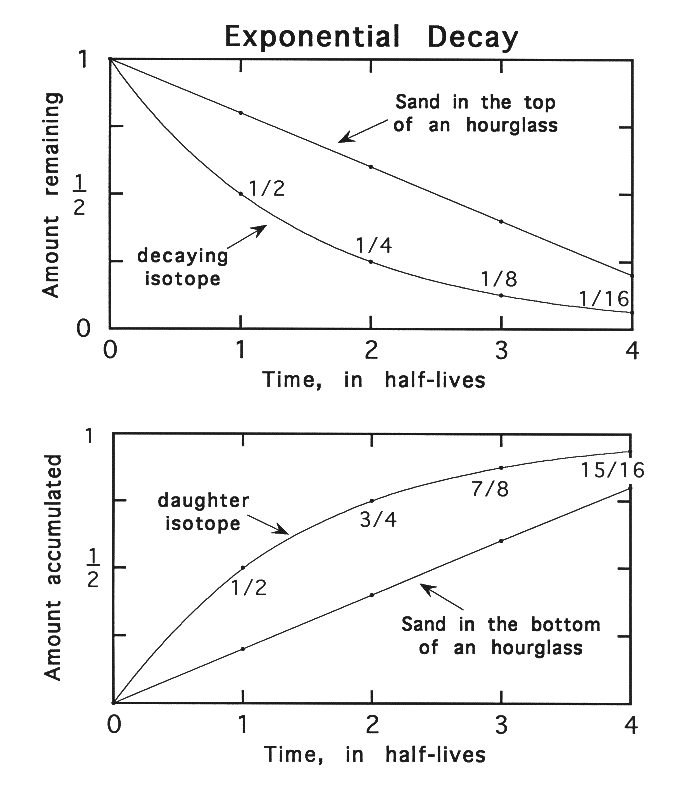
Figure 1. The rate of loss of
sand from the top of an hourglass compared to the exponential type of decay of
radioactive elements. Most processes we are familiar with are linear, like
sand in the hourglass. In exponential decay, the amount of material decreases
by half during each half-life. After two half-lives only one-fourth is left,
after three half-lives only one eighth is left, etc. As shown in the bottom
panel, the daughter element or isotope amount increases rapidly at first, then
more slowly with each succeeding half-life.
An hourglass will tell time correctly only if it is completely sealed. If
it has a hole allowing the sand grains to escape out the side instead of going
through the neck, it will give the wrong time interval. Similarly, a rock that
is to be dated must be sealed against loss or addition of either the radioactive
daughter or parent. If it has lost some of the daughter element, it will give an
inaccurately young age. As will be discussed later, most dating techniques have
very good ways of telling if such a loss has occurred, in which case the date is
thrown out (and so is the rock!).
An hourglass measures how much time has
passed since it was turned over. (Actually it tells when a specific amount of
time, e.g., two minutes, an hour, etc., has passed, so the analogy is not quite
perfect.) Like the hourglass, radiometric dating of rocks tells how much time
has passed since some event occurred. For igneous rocks the event is usually its
cooling and hardening from magma or lava. For some other materials, the event is
the end of a period of metamorphic heating (in which the rock gets baked
underground at generally over a thousand degrees Fahrenheit). The event in other
cases can be the uncovering of a surface by the scraping action of a glacier,
the chipping of a meteorite off an asteroid, or the length of time a plant or
animal has been dead.
The Radiometric Clocks
There are now well over forty different radiometric dating techniques,
each based on a different radioactive isotope.2 A partial
list of the parent and daughter isotopes and the decay half-lives is given in
Table 1. Notice the large range in the half-lives. Isotopes with long half-lives
decay very slowly, and so are useful for dating correspondingly ancient events.
Isotopes with shorter half-lives cannot date very ancient events because all of
the atoms of the parent isotope would have already decayed away, like an
hourglass left sitting with all the sand at the bottom. Isotopes with relatively
short half-lives are useful for dating correspondingly shorter intervals, and
can usually do so with greater accuracy, just as you would use a stopwatch
rather than a grandfather clock to time a 100-meter dash. On the other hand, you
would use a calendar, not a clock, to record time intervals of several weeks or
more.
Table 1. Some Naturally-Occurring Radioactive Isotopes and
Their Half-Lives
Radioactive Isotope
(Parent) |
Product
(Daughter) |
Half-Life
(Years) |
| Samarium - 147 |
Neodymium - 143 |
106 billion |
| Rubidium-87 |
Strontium-87 |
48.8 billion |
| Rhenium-187 |
Osmium-187 |
42 billion |
| Lutetium-176 |
Hafnium-176 |
38 billion |
| Thorium-232 |
Lead-208 |
14 billion |
| Uranium-238 |
Lead-206 |
4.5 billion |
| Potassium-40 |
Argon-40 |
1.26 billion |
| Uranium-235 |
Lead-207 |
0.7 billion |
| Beryllium-10 |
Boron-10 |
1.52 million |
| Chlorine-36 |
Argon-36 |
300000 |
| Carbon-14 |
Nitrogen-14 |
5715 |
Half-lives taken from N. E. Holden, Pure
Appl. Chem. 62 (1990): 941-958.
The half-lives have all been measured directly, either by using a radiation
detector to count the number of atoms decaying in a given amount of time from a
known amount of the parent material, or by measuring the ratio of daughter to
parent atoms in a sample that originally consisted completely of parent atoms.
Work on radiometric dating first started shortly after the turn of the century,
but progress was relatively slow before the late forties. For many of the dating
techniques, we now have had fifty years over which to measure and remeasure the
half-lives. Very precise counting of the decay events or the daughter atoms can
be done, so that while the number of, for example, rhenium-187 atoms decaying in
50 years is a very small fraction of the total, the resulting osmium-187 atoms
can be very precisely counted.
The uncertainties on the half-lives given
in the table are all very small. All the half-lives are known to better than
plus/minus about two percent except for rhenium (5%), lutetium (3%), and
beryllium (3%). There is no evidence of any of the half-lives changing over
time, and such a thing is forbidden by the laws of physics.
Examples of Dating Methods for Igneous
Rocks
Igneous rocks are good candidates for dating. Recall that for igneous
rocks the event being dated is when the rock was formed from magma or lava. When
the molten material cools and hardens, the atoms are no longer free to move
about. Any daughter atoms from radioactive decays occurring after the rock cools
are trapped where they are made within the rock. These atoms are like the sand
grains accumulating in the bottom of the hourglass. To determine the age of the
rock one needs to measure the number of daughter atoms and the number of
remaining parent atoms, and use the half-life to calculate the time it took to
make those daughter atoms.
However, there is one complication. One cannot
always assume that there were no daughter atoms to begin with. It turns out that
there are some cases where one can make that assumption quite reliably. But
usually the initial amount of the daughter product must be accurately
determined. Most of the time one can use the different amounts of parent and
daughter atoms present in different minerals within the rock to tell how much
daughter product was originally present. Each dating mechanism deals with this
problem in its own way. Some types of dating work better in some rocks; others
are better in other rocks, depending on the rock composition and its age. Let's
examine some of the different dating mechanisms now.
Potassium-Argon. Potassium is an abundant element in the
Earth's crust. One isotope, potassium 40, is radioactive and decays to two
different daughter products, calcium-40 and argon-40, by two different decay
methods. This is not a problem because the production ratio of these two
daughter products is precisely known, and is always constant: 11.2% becomes
argon-40 and 88.8% becomes calcium-40. It is possible to date some rocks by the
potassium-calcium method, but this is not often done because it is hard to
determine how much calcium was initially present. Argon, on the other hand, is a
gas. Whenever rock is melted to become magma or lava, the argon tends to escape.
Once the molten material hardens, it again begins to trap the argon produced
from its potassium. In this way the potassium-argon clock is clearly reset when
an igneous rock is formed.
In its simplest form, the geologist simply
needs to measure the relative amounts of potassium-40 and argon-40 to date the
rock. The age is given by a relatively simple equation:
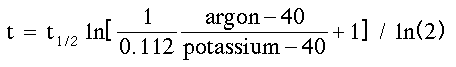
where t1/2 is the half-life, and ln is the natural
logarithm.
However, in reality there is often a small amount of argon
remaining in a rock when it hardens. This is usually trapped as very tiny air
bubbles in the rock. One percent of the air we breathe is argon. Argon from air
bubbles may need to be allowed for if it is significant compared with the amount
of radiogenic argon. This would most likely be the case in either young rocks
that have not had time to produce much radiogenic argon, or in rocks that are
not abundant in potassium. One must have a way to determine how much air-argon
is in the rock. This is rather easily done because air-argon has a couple of
other isotopes, the most abundant of which is argon-36. The ratio of argon-40 to
argon-36 in air is well known, at 295. Thus, if one measures argon-36 and
argon-40, one can calculate and subtract off the air argon-40 to get an accurate
age.
One of the best ways of showing that an age-date is correct is to
confirm it with one or more different dating technique(s). Although
potassium-argon is one of the simplest dating methods, there are still
some cases where it does not agree with other methods. When this does
happen, it is usually because the gas within bubbles in the rock is from deep
underground rather than from the air. This gas can have a higher concentration
of argon-40 escaping from the melting of older rocks. This is called
parentless argon-40 because its parent potassium is not in the rock being
dated, and is also not from the air. In these slightly unusual cases, the date
given by the normal potassium-argon method is too old. However, scientists in
the mid-1960s came up with a way around this problem, the argon-argon
method.
Argon-Argon. Although it has been around
for over a quarter of a century, the argon-argon method is seldom discussed by
groups critical of dating methods. This method uses exactly the same parent and
daughter isotopes as the potassium-argon method. In effect, it is a different
way of telling time from the same clock. Instead of simply comparing the total
potassium with the non-air argon in the rock, this method can tell exactly what
and how much argon is directly related to the potassium in the rock.
In
the argon-argon method, the rock is placed near the center of a nuclear reactor
for a number of hours. A nuclear reactor emits a very large number of neutrons,
which can change a small amount of the potassium-39 into argon-39. Argon-39 is
not found in nature because it has a 269-year half-life (the shortness of this
half-life doesn't affect the argon-argon dating method as long as the
measurements are made within about five years of the neutron dose). The rock is
then heated in a furnace to release both the argon-40 and the argon-39
(representing the potassium) for analysis. The heating is done at incrementally
higher temperatures and at each step the ratio of argon-40 to argon-39 is
measured. If the argon-40 is from decay of potassium within the rock, it will
come out at the same temperatures as the potassium-derived argon-39 and in a
constant proportion. If there is some excess argon-40 in the rock, it will
cause a different ratio of argon 40 to argon-39.
Figure 2 is an example
of a good argon-argon date. The fact that this plot is flat shows that
essentially all of the argon-40 are from decay of potassium within the rock. The
potassium-40 in the sample is found by multiplying the argon-39 by a factor
based on the neutron exposure in the reactor. When this is done, the plateau in
the figure represents an age date based on the decay of potassium-40 to
argon-40.

Figure 2. A typical argon-argon
dating plot. Each small rectangle represents the apparent age given at one
particular heating-step temperature. The top and bottom parts of the
rectangles represent upper and lower limits for that particular determination.
The x-axis gives the amount of the total argon-39 released from the sample. A
good argon-argon age determination will have many heating steps which all
agree with each other. The "plateau age" is the age given by the average of
most of the steps; in this case it is 136.4 million years. The sample is a
Parana continental flood basalt from Brazil. From S. Turner, et al., Earth
and Planetary Science Letters 121 (1994): 333-348.
There are occasions when the argon-argon dating method does not give an
age even if there is sufficient potassium in the sample and the rock was old
enough to date. This most often occurs if the rock experienced a high
temperature (usually a thousand degrees Fahrenheit or more) at some point since
its formation. If that occurs, some of the argon gas moves around, and the
analysis does not give a smooth plateau across the extraction temperature steps.
An example of an argon-argon analysis which did not yield an age date is shown
in Figure 3. Notice that there is no good plateau in this plot. Sometimes, there
will actually be two plateaus, one representing the formation age, and another
representing the time at which the heating episode occurred. Usually where the
system has been disturbed, there simply is no date given. The important point to
note is that, rather than giving wrong age dates, this system simply does not
give a date if the system has been disturbed. This is also true of several
other igneous rock dating methods, as we will describe below.

Figure 3. An argon-argon plot
which gives no date. Note that the apparent age is different for each
temperature step so that there is no plateau. This sample was struck with a
pressure of 420,000 atmospheres to simulate a meteorite impact--an extremely
rare event. The impact heated the rock and caused its argon to be rearranged,
so that it could not give an argon argon date. Before it was smashed, the rock
gave an age of around 450 million years, as shown by the dotted line. From A.
Deutsch and U. Schaerer, Meteoritics 29 (1994):
301-322.
Rubidium-Strontium. In nearly all of the dating
techniques, except potassium-argon and the associated argon-argon method, there
is always some amount of the daughter product already in the rock when it cools.
Using these methods is a little like trying to tell time from an hourglass that
was turned over before all of the sand had fallen to the bottom. One can think
of ways to correct for this in an hourglass: One could make a mark on the
outside of the glass where the sand level started from and then repeat the
interval with a stopwatch in the other hand to calibrate it. Or if one is clever
she or he could examine the hourglass's shape and determine what fraction of all
the sand was at the top to start with. By knowing how long it takes all of the
sand to fall, one could determine how long the time interval was. Similarly,
there are good ways to tell quite precisely how much of the daughter product was
already in the rock when it cooled and hardened.
In the
rubidium-strontium method, rubidium-87 decays with a half-life of 48.8 billion
years to strontium 87. Strontium has several other isotopes that are stable and
do not decay. The ratio of strontium-87 to another stable isotope, say
strontium-86, increases over time as more rubidium-87 turns to strontium-87. But
when the rock first cools, all parts of the rock have the same
(strontium-87/strontium-86) ratio because the isotopes were mixed in the magma.
At the same time, some minerals in the rock have a higher rubidium/strontium
ratio than others. Rubidium has a larger atomic diameter than strontium, so that
rubidium does not fit into the crystal structure of some minerals as well as it
does in others.
Figure 4 is an important type of plot used in
rubidium-strontium dating. It shows the strontium 87/strontium-86 ratio on the
vertical axis and the rubidium-87/strontium-86 ratio on the horizontal axis,
that is, it plots a ratio of the daughter isotope against a ratio of the parent
isotope. At first, all the minerals lie along a horizontal line of constant
strontium-87/strontium-86 but with varying rubidium/strontium. As the rock
starts to age, rubidium gets converted to strontium. The amount of strontium
added to each mineral is proportional to the amount of rubidium present. This
change is shown by the dashed arrows, the lengths of which are proportional to
the rubidium/strontium ratio. The dashed arrows are slanted because the
rubidium/strontium ratio is decreasing in proportion to the increase in
strontium-87/strontium-86. The solid line drawn through the samples will thus
progressively rotate from the horizontal to steeper and steeper slopes.

Figure 4. A rubidium-strontium
three-isotope plot. When a rock cools, all its minerals have the same ratio of
strontium 87 to strontium-86, though they have varying amounts of rubidium. As
the rock ages, the rubidium decreases by changing to strontium-87, as shown by
the dotted lines. Minerals with more rubidium gain more strontium-87, while
those with less rubidium do not change as much. At any given time the
composition can be represented as a line through the points. Notice that the
line can be extrapolated back to zero to give the initial
strontium-87/strontium-86 ratio. The age of the rock can be determined from
the slope of the line.
A line drawn through the samples at any later time will intersect the
horizontal line at the same point in the lower left-hand corner. This point,
where rubidium-87/strontium-86 equals zero, tells the original
strontium-87/strontium-86 ratio. From that we can determine the original
daughter strontium-87 in each mineral, which is just what we need to know to
determine the correct age.
It also turns out that the slope of the line
is proportional to the age of the rock: the older the rock, the steeper the
line. If the slope of the line is m and the half-life is t1/2, the age t (in
years) is given by the equation

For a system with a very long half-life like rubidium-strontium, the actual
numerical value of the slope will always be quite small. To give an example for
the above equation, if the slope of a line in a plot similar to Fig. 4 is m =
0.05110 (strontium isotope ratios are usually measured very accurately--to about
one part in ten thousand), we can substitute in the half-life (48.8 billion
years) and solve as follows:
|
t = (48.8) x ln(1.05110)/ln(2) |
| so |
t = 3.51 billion years. |
There are several
things that, on rare occasions, can cause problems for the rubidium-strontium
dating method. One possible source of problems is if a rock contains some
minerals that are older than the main part of the rock. Sometimes magma inside
the earth will pick up unmelted minerals from the surrounding rock as it moves
through a magma chamber. Usually a good geologist can distinguish these
"xenoliths" from the younger minerals all around them. If he or she does happen
to use them for dating the rock, the points represented by these minerals will
lie off the line made by the rest of the points. Other difficulties arise if a
rock has undergone metamorphism, that is, if the rock got very hot, but not hot
enough to completely remelt the rock. In these cases, the dates look confused,
and do not lie along a line. Some minerals may have completely melted, while
others did not melt at all, so that some minerals try to give the igneous age
while other minerals try to give the metamorphic age. In these cases there will
not be a straight line, and no date is determined.
In a few, very
rare instances, the rubidium-strontium method has given straight lines that give
wrong ages. This can happen when the rock being dated was formed from magma that
was not well mixed, and which had two distinct batches of rubidium and
strontium. One magma batch had rubidium and strontium compositions near the
upper end of a line (such as in Fig. 4), and one batch had compositions near the
lower end of the line. In this case, the minerals all got a mixture of these two
batches, and their resulting composition ended near a line between the two
batches. This is called a two-component mixing line. It is a very rare
occurrence in these dating mechanisms, but at least thirty cases have been
documented among the tens of thousands of rubidium-strontium dates made. If a
two-component mixture is suspected, a second dating method must confirm or
disprove the rubidium-strontium date. The agreement of several dating methods is
the best fail-safe way of dating rocks.
The
Samarium-Neodymium, Lutetium-Hafnium, and Rhenium-Osmium Methods. All these
methods work very similarly to the rubidium-strontium method. They all use
three-isotope diagrams similar to Figure 4 to determine the age. The
samarium-neodymium method is the most often-used of these three. It uses the
decay of samarium-147 to neodymium-143, which has a half life of 105 billion
years. The ratio of the daughter isotope, neodymium-143, is plotted against the
ratio of the parent, samarium-147, with neodymium-144 in the denominators. If
different minerals from the same rock plot along a line, the slope is
determined, and the age is given by the same equation as above. The
samarium-neodymium method may be preferred for rocks that have very little
potassium and rubidium, which would make dating by the potassium-argon,
argon-argon, and rubidium-strontium methods difficult. The samarium-neodymium
method has also been shown to be more resistant to being disturbed by
metamorphic heating events, so for some metamorphosed rocks the
samarium-neodymium method is preferred. For a rock of the same age, the slope on
the neodymium-samarium plots will be less than on a rubidium-strontium plot
because the half-life is longer. However, these isotope ratios are usually
measured to extreme accuracy--several parts in ten thousand--so that accurate
dates can be obtained even for ages less than one fiftieth of a half-life, and
correspondingly small slopes.
The lutetium-hafnium method uses the 38
billion year half-life of lutetium-176 decaying to hafnium-176. This dating
system is similar in many ways to samarium-neodymium, as the elements tend to be
concentrated in the same types of minerals. Since samarium-neodymium dating is
somewhat easier, the lutetium-hafnium has not been used in very many
cases.
The rhenium-osmium method takes advantage of the fact that the
osmium concentration in most rocks and minerals is very low, so that a small
amount of the parent rhenium-187 can produce a significant change in the osmium
isotope ratio. The half-life for this radioactive decay is 42 billion years. The
nonradiogenic stable isotopes, osmium-186 or -188, are used as the denominator
in the ratios on the three-isotope plots. This method has been useful for dating
iron meteorites, and is now enjoying greater use for dating earth rocks due to
development of easier rhenium and osmium isotope measurement
techniques.
Uranium-Lead and related techniques.
The uranium-lead method is the longest-used dating method. It was first used in
1907, some ninety years ago. The uranium-lead system is more complicated than
other parent-daughter systems; it is actually several dating methods put
together. Natural uranium consists primarily of two isotopes, U-235 and U-238,
and these isotopes decay with different half-lives to produce lead-207 and
lead-206, respectively. In addition, lead-208 is produced by thorium-232. Only
one isotope of lead, lead-204, is not radiogenic. The uranium-lead system has an
interesting complication: none of the lead isotopes is produced directly from
uranium and thorium. Each decays through a series of short-lived radioactive
elements that are produced and almost immediately decay to a lighter element,
finally ending at lead. Since these half-lives are so short compared to uranium
and thorium, they do not affect the overall dating scheme. The result is that
one can obtain three independent estimates of the age of a rock by measuring the
lead isotopes and their parent isotopes, uranium-235 and -238 and
thorium-232.
The uranium-lead system in its simpler forms has proved to
be less reliable than many of the other dating systems. This is because both
uranium and lead are less easily retained in many minerals in which they are
found. The intermediate elements in the decay chain do not help matters either.
Yet the fact that there are three dating systems all in one allows scientists to
easily determine whether the system has been disturbed or not. Using slightly
more complicated mathematics, different combinations of the lead isotopes and
parent isotopes can be plotted in a way that minimizes the effects of lead loss.
One of these techniques is called the lead-lead technique because it determines
the ages from the lead isotopes alone. Some of these techniques allow scientists
to chart at what points in time metamorphic heating events have occurred. This
information is also of significant interest to geologists.
Cosmogenic Radionuclides:
Carbon-14,
Beryllium-10, Chlorine-36
The last three radiometric systems listed in Table 1 have far shorter
half-lives than all the rest. Unlike the other radioactive isotopes, carbon-14,
beryllium-10, and chlorine-36 are constantly being replenished in small amounts
by a special mechanism. Cosmic rays--high energy particles and photons in
space--produce these isotopes in air, very high in the earth's atmosphere. Very
small amounts of each of these isotopes are present in the air we breathe and
the water we drink. As a result, living things, both plants and animals, ingest
very small amounts of carbon-14, and lake and sea sediments take up small
amounts of beryllium-10 and chlorine-36.
The cosmogenic dating clocks
work somewhat differently than the others. Carbon-14 in particular is used to
date organic material such as bones, wood, cloth, paper, and other dead tissue
from either plants or animals. To a rough approximation, the ratio of carbon-14
to the stable isotopes, carbon-12 and carbon-13, is more or less constant in the
atmosphere and living organisms. Once a living thing dies, it no longer takes in
carbon from food or air, and the amount of carbon-14 starts to drop with time.
How far the (carbon-14/carbon-12) ratio has dropped indicates how old the sample
is. Since the half-life of carbon-14 is less than 6,000 years, it can only be
used for dating material less than about 40,000 years old. Dinosaur bones do not
have carbon-14 (unless contaminated), as the dinosaurs became extinct over 60
million years ago, but some other animals that are now extinct, such as North
American mammoths, can be dated by carbon-14. Also, some materials from
prehistoric times, as well as biblical events, can be dated by
carbon-14.
The carbon-14 system has been carefully calibrated with
nonradiometric age indicators. For example growth rings in trees, if counted
carefully, are a reliable way to determine the age of a tree. Each growth ring
only collects carbon from the air and nutrients during the year it is made. To
calibrate carbon-14, one can analyze carbon from several center rings of a tree,
and then count the rings inward from the living portion to determine the actual
age. This has been done for the "Methuselah of trees," the bristlecone pines,
which grow very slowly and live up to 6,000 years. Scientists have extended this
calibration even further. These trees grow in a very dry region near the
California-Nevada border. Dead trees in this dry climate take many thousands of
years to decay. Growth ring patterns based on wet and dry years can be
correlated between living and long dead trees, extending the ring count back to
about 10,000 years ago.
There are other ways of extending farther back in
time. One of the best known is the seasonal variations in oxygen isotopes in
polar ice from Greenland and Antarctica. Because winter ice has a greater
concentration of the lighter isotope, oxygen-16, each winter's deposit makes an
invisible layer in the ice, something like a tree ring, which is detectable by
isotope analysis. This record goes back about 100,000 years.
While the
ice cores are not so easy to compare with carbon 14 dates, the tree rings are
very easy to use since trees contain a lot of carbon. The comparison of
radiocarbon ages of tree rings with their known ages has revealed variations of
up to 12% between the predicted and actual ages. The variations occur because
the source of carbon-14 at the top of the atmosphere is slightly variable. This
phenomenon is quite well understood, and affects only the clocks that rely on
cosmic ray production (e.g., carbon-14, beryllium-10, and chlorine-36).
Carbon-14 dates of less than about 10,000 years are corrected for this effect,
and there is no reason to believe that older radiocarbon dates are altered to
any larger degree. In summary, thousands of radiocarbon dates are obtained each
year on organic matter less than about forty thousand years old. It has proved
to be a reliable clock for such time scales. A research journal by the name
Radiocarbon, published since 1959, is devoted exclusively to this study
and is available in many geology and archeology libraries around the
country.
The Age of the Earth
| Some of the oldest rocks on earth are found in Western Greenland.
Because of their great age, they have been especially well studied. The
table below gives the ages, in billions of years, from twelve different
studies using five different techniques on one particular rock formation
in Western Greenland, the Amitsoq gneisses. |
| Technique |
Age Range (billion years) |
| uranium-lead |
3.60±0.05 |
| lead-lead |
3.56±0.10 |
| lead-lead |
3.74±0.12 |
| lead-lead |
3.62±0.13 |
| rubidium-strontium |
3.64±0.06 |
| rubidium-strontium |
3.62±0.14 |
| rubidium-strontium |
3.67±0.09 |
| rubidium-strontium |
3.66±0.10 |
| rubidium-strontium |
3.61±0.22 |
| rubidium-strontium |
3.56±0.14 |
| lutetium-hafnium |
3.55±0.22 |
| samarium-neodymium |
3.56±0.20 |
| (compiled from Dalrymple,
1991) | |
| Note that scientists give their results with a stated uncertainty.
They take into account all the possible errors and give a range within
which they are 95% sure that the actual value lies. The top number,
3.60±0.05, refers to the range from 3.55 to 3.65 billion years. The size
of this range is every bit as important as the actual number. One number
with a small uncertainty range is more accurate than a number with a
larger range. For the numbers given above, one can see that all of the
ranges overlap and agree between 3.62 and 3.65 billion years as the
age of the rock. Several studies also showed that, because of the great
ages of these rocks, they have been through several mild metamorphic
heating events that disturbed the ages given by potassium-bearing
minerals. |
We now turn our attention to what the dating
systems tell us about the age of the earth. The most obvious constraint is the
age of the oldest rocks. These have been dated at up to about four billion
years. But actually only a very small portion of the earth's rocks are that old.
From satellite data we know that the earth's surface is constantly rearranging
itself little by little as earthquakes occur. Such rearranging cannot occur
without some of the earth's surface disappearing under other parts of the
earth's surface, remelting some of the rock. So it appears that none of the
rocks have survived from the creation of the earth without undergoing remelting,
metamorphism, or erosion, and all we can say--from this line of evidence--is
that the earth appears to be at least as old as the four billion-year-old rocks.
When scientists began systematically dating meteorites, they learned a
very interesting thing: nearly all of the meteorites had practically identical
ages, at 4.56 billion years. These meteorites are chips off the asteroids. When
the asteroids were formed in space, they cooled relatively quickly (some of them
may never have gotten very warm), so all of their rocks were formed within a few
million years. The asteroids' rocks have not been remelted ever since, so the
ages have generally not been disturbed. Meteorites which show evidence of being
from the largest asteroids have slightly younger ages. The moon is larger than
the largest asteroid. The oldest rocks we have from the moon do not exceed 4.1
billion years, though a larger sampling might yield some slightly older ages.
Most scientists think that all the bodies in the solar system were created about
the same time. There is evidence from the uranium, thorium, and lead isotopes
that links the earth's age with that of the meteorites. This would make the
earth about 4.5-4.6 billion years old.
Notice one other important detail
about radioactive isotopes. Most of the naturally-occurring radioactive isotopes
mentioned above have very long half-lives, on the order of billions of years.
The only ones with shorter half-lives are those which have a source constantly
replenishing them, such as the carbon-14, beryllium-10 and chlorine-36 produced
by cosmic rays. We can make hundreds of other radioactive isotopes with
half-lives shorter than a billion years, but they do not occur naturally on
earth. Occasionally there is evidence that these isotopes existed at some point
in the past, but have since decayed completely away. The longest half-lives of
this group of "extinct" radionuclides are close to a hundred million years. Why
do we find almost no short-lived radionuclides and so many long-lived ones? This
is what one would expect to find if God had created the earth approximately four
and a half billion years ago. The earth is old enough that radioactive isotopes
with half-lives of up to a hundred million years decayed away, but not so old
that radioactive isotopes with half-lives close to a billion years are gone.
Can We Really Believe the Dating
Systems?
Some Christians question whether we can believe something so far back in
the past. My answer is that it is similar to believing in other things of the
past. It only differs in degree. Why do you believe Abraham Lincoln ever lived?
Because it would take an extremely elaborate scheme to make up his existence,
including forgeries, fake photos, and many other things, and besides, there is
no good reason to simply have made him up. Well, the situation is very similar
for the dating of rocks, only we have rock records rather than historical
records. Consider the following:
- There are well over forty different radiometric dating methods, and scores
of other methods such as tree rings or ice cores.
- All of the different dating methods agree--they agree a great majority of
the time over millions of years. Some Christians make it sound like there is
much disagreement, but this is not so. The disagreement in values needed to
support the position of young-earth creationists would require differences
measured by orders of magnitude (e.g., factors of 1000, 10,000, a million, or
more). The differences actually found in the scientific literature are usually
close to the margin of error, not orders of magnitude.
- There are vast amounts of data overwhelmingly favoring the old earth
model. There are several hundred laboratories around the world actively doing
radiometric dating; all but possibly one or two favor the old earth. A quick
word search of the word "dating" shows that among the Caltech library
holdings, in 1994 alone there were at least 450 research papers published
specifically on dating of geologic and archaeologic materials. At least 95 of
these were on rubidium-strontium, and 42 were on potassium-argon or
argon-argon. Essentially all these favored an old earth.
- Radioactive decay rates have been measured for over forty years now for
many of the decay clocks without any observed changes. And it has been over
eighty years since the uranium decay rate was first determined.
- The mathematics for determining the ages from the observations is
relatively simple.
The last two points deserve more attention. Some Christians have argued
that something may be slowly changing with time so that all the ages look older
than they really are. The only two quantities in the exponent of a decay rate
equation are the half-life and the time. So for ages to appear longer than
actual, all the half-lives would have to be changing in sync with each other.
One could consider that time itself was changing if that happened (Remember that
our clocks are now standardized to atomic clocks!). And such a thing would have
to have occurred without our detection in the last 80 years, which is already 4%
of the way back to the time of Christ.
It would not be inconsistent with
the scientific evidence to conclude that God made everything relatively
recently, but with the appearance of great age, just as Genesis 1 and 2 tell of
God making Adam as a fully grown man (which implies the appearance of age). That
is a philosophical and theological matter which we won't go into here, though it
has some shades of the Abraham Lincoln example. We only note here that an
apparent old earth is consistent with the great amount of scientific
evidence.
Doubters Still Try
Some doubters have tried to dismiss geologic dating with a sleight of
hand by saying that no rocks are completely closed systems (that is, that no
rocks are so isolated from their surroundings that they have not lost or gained
some of the isotopes used for dating). Speaking from an extreme technical
viewpoint this might be true--perhaps one atom out of one trillion of a certain
isotope has leaked out of nearly all rocks, but such a change would make an
unmeasurably small change in the result. The real question to ask is: "Is the
rock sufficiently close to a closed system that the results will be the same as
a really closed system?" Since the early 1960s many books have been written on
this subject. These books detail experiments showing, for a given dating system,
which minerals work all of the time, which minerals work under some certain
conditions, and which minerals are likely to lose atoms and give incorrect
results. Understanding these conditions is part of the science of geology.
Geologists are careful to use the most reliable methods whenever possible, and
as discussed above, to test for agreement between different methods.
Some
people have tried to defend a young earth position by saying that the half-lives
of radionuclides can in fact be changed, and that this can be done by certain
little-understood particles such as neutrinos, muons, or cosmic rays. This is
stretching it. While certain particles can cause nuclear changes, they do not
change the half-lives. The nuclear changes are well understood and are nearly
always very minor in rocks. In fact the main nuclear changes in rocks are the
very radioactive decays we are talking about.
There are only three quite
technical instances where a half-life changes, and these do not affect the
dating techniques we have discussed.
1. According to theory, a certain
type decay called electron-capture decay is most likely to show changes with
pressure or chemical combination, and that should be most pronounced for very
light elements. The synthetic isotope, beryllium-7, has indeed been shown by
several people to change by up to a fraction of a percent. In one such
experiment, beryllium-7 was subjected to 270,000 atmospheres of pressure, which
would only occur at depths greater than 450 miles inside the earth. All known
rocks, with the possible exception of diamonds, are from much shallower depths.
In fact, beryllium-7 is not used for dating rocks, as it has a half-life of only
54 days, and heavier nuclei are even less subject to these minute changes, so
that the dates of rocks made by electron-capture decays would only be off by at
most a few hundredths of a percent.
2. Cosmic rays are very, very
high-energy atomic nuclei flying through space. The electron-capture decay
mentioned above does not take place in cosmic rays until they slow down. This is
because the fast moving cosmic rays do not have electrons. All normal matter,
such as everything on earth, the moon, meteorites, etc. always has electrons, so
this instance does not affect things we date.
3. The last case also
involves very fast-moving matter. It has been demonstrated by atomic clocks in
rapid very fast spacecraft. These atomic clocks slow down very slightly (only a
second or so per year) as predicted by Einstein's theory of relativity. No rocks
in our solar system are going fast enough to make a noticeable change in their
dates.
These cases are very specialized, and all are well understood.
None of these cases alter the dates of rocks either on earth or other places in
the solar system. The conclusion again is that half-lives are completely
reliable in every context, including the dating of rocks on earth and even on
other planets.
Rightly Handling the Word of Truth
As Christians it is very important that we understand God's Word
correctly. Yet from the middle ages until the 1700s, people insisted that the
Bible taught that the earth, not the sun, was the center of the solar system. It
wasn't that people just thought it had to be that way; they actually quoted
Scriptures: "The earth is firmly fixed; it shall not be moved" (Psalm
104:5), or "the sun stood still" (Joshua 10:13; why should it say the sun
stood still if it is the earth's rotation that causes day and night?),
and many other passages. I am afraid the debate over the age of the earth has
many similarities. But I am optimistic. Today there are many Christians who
accept the reliability of geologic dating, but do not compromise the spiritual
and historical inerrancy of God's word. While a full discussion of Genesis 1 is
not given here, references are given below to books which deal with that
issue.
As scientists, we deal daily with what God has revealed about
himself through the created universe. The psalmist marveled at how God, Creator
of the universe, could care about humankind: "When I consider Your heavens,
the work of Your fingers, the moon and the stars, which You have set in place,
what is man that You are mindful of him, the son of man that You care for
him?" (Psalm 8:3-4). Near the beginning of the twenty-first century we can
marvel even more, knowing how vast the universe is, how ancient the rocks and
hills are, and how carefully our environment has been designed. Truly God is
more awesome than we can imagine!
APPENDIX
Common Misconceptions
Regarding
Radiometric Dating Techniques
There are several misconceptions that seem especially prevalent among
Christians. Most of these topics are covered in the above discussion, but they
are reviewed briefly here for clarity.
1. Radiometric dating is based
on index fossils.
This is not at all true, though it has actually
been suggested. Radiometric dating is based on the half-lives of the radioactive
isotopes. These half-lives have been measured over the last 40-80 years. They
are not calibrated at all by fossils.
2. The decay rates are poorly
known, so the dates are inaccurate.
Most of the decay rates used for
dating rocks are known to within 2 percent. Uncertainties are only slightly
higher on rhenium (5%), lutetium (3%), and beryllium (3%). Such small
uncertainties are no reason to dismiss radiometric dating. Whether a rock is 100
million years or 102 million years old does not make a great deal of
difference.
3. A small error in the half-lives leads to a very large
error in the date.
Since exponents are used in the dating equations,
it is possible for people to think this might be true, but it is not. If a
half-life is off by 2%, it will only lead to a 2% error in the date.
4.
Decay rates can be affected by the physical surroundings.
This is
not true in dating rocks. Radioactive atoms used for dating have been subjected
to heat, cold, pressure, vacuum, acceleration, and strong chemical reactions
without any measurable change. The only exceptions, which are not relevant to
dating rocks, are discussed under the section, "Doubters Still Try,"
above.
5. No one has measured the decay rates directly; we only know
them from inference.
Decay rates have been directly measured over the
last 50-80 years. In some cases a batch of the pure parent material is weighed
and set aside for a long time, and then the resulting daughter material is
weighed. In many cases it is easier to detect radioactive decays by the energy
burst each decay gives off. For this, a batch of the pure parent material is
carefully weighed and then put in front of a Geiger counter which counts the
number of decays over a long time.
6. The decay rates might be slowing
down over time, leading to incorrect old dates.
While we cannot rule
out that this could possibly have happened in the past, there is no evidence
that anything of the sort has happened in the past century. And the following
argument makes this suggestion meaningless in terms of apparent ages: Since the
different dating methods are in good agreement, all of the half-lives must have
slowed down the same amount together. Such an occurrence would be the same as if
time itself slowed down. But everything still appears very old, so why
complicate things by making this suggestion in the first place?
7.
There is little or no way to tell how much of the decay product was
originally in the rock, leading to anomalously old ages.
A good part
of this work is devoted to explaining how one can tell how much of a given
element or isotope was originally present. Usually it involves using more than
one sample from a given rock. By comparing the ratios of parent and daughter
isotopes relative to a stable isotope for samples with different relative
amounts of the parent isotope, one can determine how much of the daughter
isotope would be present if there had been no parent isotope. This is the same
as the initial amount (it would not change if there was no parent isotope to
decay). Figure 4 and the accompanying explanation tell how this is done most of
the time. While this is not absolutely 100% foolproof, comparison of several
dating methods will always show whether the given date is reliable.
8.
There are only a few different dating techniques.
We have listed
eleven different radiometric dating techniques and discussed them. These make up
only the tip of the iceberg. There are over forty different radiometric dating
techniques in use, and there are many other dating techniques making use of rare
stable isotopes, yearly variations such as tree rings and ice cores, and other
reliable methods.
9. "Radiation halos" in rocks prove that the earth
was young.
This refers to tiny halos of crystal damage surrounding
spots where radioactive elements are concentrated in certain rocks. Halos
thought to be from polonium, a short-lived element produced from the decay of
uranium, have been found in some rocks. A plausible explanation for a halo from
such a short-lived element is that these were not produced by an initial
concentration of the radioactive element. Rather, as water seeped through cracks
in the minerals, a chemical change caused newly-formed polonium to drop out of
solution at a certain place and almost immediately decay there. A halo would
build up over a long period, although the center of the halo never contained
more than a few atoms of polonium at one time. "Hydrothermal" effects can act in
ways that at first seem strange, such as the well-known fact that gold--a
chemically unreactive metal with very low solubilities--is concentrated along
quartz veins by the action of water over long periods of time. Other researchers
have found halos produced by an indirect radioactive decay effect called hole
diffusion, which is an electrical effect in a crystal. These results suggest
that the halos in question are not from short-lived isotopes after
all.
At any rate, halos from uranium inclusions are far more common.
Because of uranium's long half-lives, these halos take at least several hundred
million years to form. Because of this, most people agree that halos provide
compelling evidence for a very old earth.
10. Only atheists and
liberals are involved in radiometric dating.
The fact is that there
are many Bible-believing Christians who are involved in radiometric dating, and
who can see its validity firsthand. Most of the members of the Affiliation of
Christian Geologists are firmly convinced that radiometric dating shows evidence
that God created the earth billions, not thousands, of years ago.
11.
Different dating techniques usually give conflicting results.
This
is not true at all. The fact that dating techniques most often agree with each
other is why scientists tend to trust them in the first place. Nearly every
college and university library in the country has periodicals such as
Science, Nature, and specific geology journals which give the
results of dating studies. The public is usually welcome to (and should!) browse
in these libraries. So the results are not hidden; people can go look at the
results for themselves. In 1994 alone, at least 450 research articles were
published, essentially all favoring a very old earth. Besides the scientific
periodicals which carry up-to-date research reports, specific suggestions are
given below of books for further reading.
Further Reading
- Van Till Howard J., Young Davis A., and Menninga, Clarence. (1988)
Science Held Hostage. InterVarsity, Downers Grove, IL, 189 pp.
- This book talks about the misuse of science by both hard-line atheists and
by young-earth creationists. A good deal of the book is devoted to refuting
young-earth arguments, including a substantial section on the Grand Canyon
geology. Its authors are well-known Christians in Geology and Physics.
- Ross, Hugh. (1994) Creation and Time: A Biblical and Scientific
Perspective on the Creation-Date Controversy. NavPress, Colorado Springs,
CO.
- Hugh Ross has a Ph.D. in Astronomy and he defends modern science and an
old age for the universe, refutes common young-earth arguments, and firmly
believes in the inerrancy of the Bible.
- Young, Davis A. (1982) Christianity and the Age of the Earth.
Zondervan, Grand Rapids, MI (now available through Artisan Sales, Thousand
Oaks, CA).
- Davis Young has a Ph.D. in Geology and teaches at Calvin College. He
argues for an old earth and refutes many of the common young-earth claims
(including their objections to radiometric dating).
- Strahler, Arthur N. (1987) Science and Earth History: The
Evolution/Creation Controversy. Prometheus Books, Buffalo, pp. 129-138.
- This book was not written by a Christian, but is included here because it
is a very thorough and comprehensive refutation of young-earth evidences. The
only negative aspect is that at one point Strahler throws in a bit of his own
theology--his arguments against the need for a God. This book is long and in
small print; it covers a wealth of information.
- Wiester, John. (1983) The Genesis Connection. Interdisciplinary
Biblical Research Institute, Hatfield, PA, 254 pp.
- John Wiester has taught Geology at Westmont and Biola University, and is
active in the American Scientific Affiliation, an organization of scientists
who are Christians. This book discusses many scientific discoveries relating
to the age of the earth and how these fit into the context of Genesis 1.
Stoner, Don. (1992) A New Look at an Old Earth. Schroeder,
Paramount, CA, 191 pp.
- A persuasive book written for the Christian layman. Stoner uses arguments
both from the theological and the scientific side. He talks somewhat
philosophically about whether God deceives us with the Genesis account if the
earth is really old. Stoner also tries to discuss the meaning of the Genesis 1
text.
Dickin, Alan. (1995) Radiometric Isotope Geology. Cambridge
University Press, 452 pp.
- This is the most up-to-date and comprehensive textbook on radiometric
dating techniques.
The following books are popular college-level Geology texts that deal in
depth with various dating techniques. Geologic Time is very easy to read
and has been around for some time. The text by Dalrymple is meant to be
relatively easy to read, but is also very comprehensive. The Faure texts are
regular textbooks for Geology, including more mathematics and more details.
- Dalrymple, G. Brent. (1991) The Age of the Earth. Stanford
University Press, 474 pp.
Eicher, Don L. (1976) Geologic Time, 2nd edition. Prentice-Hall,
Englewood Cliffs, NJ, 150 pp.
Faure, Gunter. (1986) Principles of
Isotope Geology, 2nd edition. Wiley, New York, 464 pp.
- Faure, Gunter. (1991) Principles and Applications of Inorganic
Geochemistry: A Comprehensive Textbook for Geology Students. MacMillan
Pub. Co., New York, 626 pp.
Electronic media: There is a site on the world wide web called
Talk.Origins at "http://rumba.ics.uci.edu:8080/". Information on radiometric
dating can be found under several FAQ (frequently asked questions) subheadings.
The information can also be accessed by anonymous ftp to
"ics.uci.edu:/pub/origins".
American Scientific
Affiliation
http://ursa.calvin.edu/chemistry/ASA/
Affiliation of
Christian Geologists
http://www.ksu.edu/~kbmill/acghome.html
Science
& Christianity Resource
List
http://hercules.geology.uiuc.edu/~Schimmri/christianity/scichr
Reasons
to
Believe
http://www.reasons.org/reasons/index.html
Acknowledgments:
Several members of the Affiliation of Christian Geologists and other Christians
involved in the sciences reviewed this paper and/or made contributions. The
following people are sincerely thanked for their contributions: Drs. Jeffery
Greenberg and Stephen MosHier (Wheaton College), John Wiester (Westmont
College), Dr. Davis Young (Calvin College), Dr. Elaine Kennedy (Loma Linda
University), Steven Schimmrich (U. of Illinois), Kenneth VanDellen (Macomb
Community College), Dr. Guillermo Gonzalez (U. Texas, Austin), Ronald Kneusel,
and James Gruetzner (U. New Mexico).
About the author: Dr.
Wiens received a bachelor's degree in Physics at Wheaton College and his Ph.D.
at the University of Minnesota doing research on meteorites and moon rocks. He
spent two years at Scripps Institution of Oceanography (La Jolla, CA) where he
studied isotopes of helium, neon, argon, and nitrogen in terrestrial rocks. He
is presently a staff scientist in the Geological and Planetary Sciences Division
at Caltech, where he is continuing to study meteorites and is involved in a
space mission to return to earth a sample of solar wind. He has published over a
dozen scientific research papers and has also published articles in Christian
magazines. Dr. Wiens became a Christian at a young age, and has been a member of
Mennonite Brethren, General Conference Baptist, and Conservative Congregational
churches.
GLOSSARY
- Atom The smallest unit that materials can be divided into. An atom
is about ten billionths of an inch in diameter and consists of a nucleus of
nucleons (protons and neutrons) surrounded by electrons.
- Closed system A system (rock, planet, etc.) which has no influence
or exchange with the outside world. In reality there is always some exchange
or influence, but if this amount is completely insignificant for the process
under consideration (e.g., for dating, if the loss or gain of atoms is
insignificant) for practical purposes the system can be considered closed.
- Cosmic rays Very high energy particles which fly through space.
They are stopped by the earth's atmosphere, but in the process, they
constantly produce carbon-14, beryllium-10, chlorine-36, and a few other
radioactive isotopes in small quantities.
- Cosmogenic Produced by bombardment of cosmic rays. Carbon-14 is
said to be cosmogenic because it is produced by cosmic
rays.
Daughter The element or isotope which is produced by
radioactive decay.
- Decay The change from one element or isotope to another. Only
certain isotopes decay. The rest are said to be stable.
- Electron-capture decay The only type of radioactive decay that
requires the presence of something--an electron--outside the atom's nucleus.
Electron capture decay of light atoms--those having the fewest electrons--can
be very slightly affected by extremely high pressures or certain chemical
bonds, so as to change their half-lives by a fraction of a percent. But no
change in the half-lives of elements used for radiometric dating has ever been
verified.
- Element A substance that has a certain number of protons in the
nucleus and unique properties. Elements may be further broken down into
isotopes, which have nearly all of the same properties except for their mass
and their radioactive decay characteristics.
Half-life The amount of
time it takes for half the atoms of a radioactive isotope to decay.
- Ice core Long sections of ice brought up by special drilling rigs
on the ice sheets of Greenland and Antarctica.
- Igneous rock Rock formed from molten lava. The other two types of
rock are sedimentary, formed by the cementing together of soil or sand, and
metamorphic rocks reformed by heat over long periods of time.
- Isotope Atoms of a given element that have the same atomic mass.
Most elements have more than one isotope. Most radioactive elements used for
dating have one radioactive isotope and at least one stable isotope. For
example carbon-14 (which weighs 14 atomic mass units) is radioactive, while
the more common isotopes, carbon-12 and carbon-13 are not.
- Lead-lead dating A variation on the uranium-lead technique in which
only the isotopes of lead need to be measured.
- Magma Hot molten material from which rocks are formed. When magma
erupts on the surface of the earth, it is called lava.
- Metamorphism The heating of rocks over long time periods at
temperatures which are hot enough to change the crystal structure but not hot
enough to completely melt the rock. Metamorphism tends to alter or reset the
radiometric time clocks, though some radiometric techniques are more resistant
to resetting than others.
Nucleons Neutrons and protons, which make
up the nucleus of an atom.
Parent The element or isotope which
decays. The element it produces is called the daughter.
- Radioactive Subject to change from one element to another. During
the change, or decay, energy is released either as light or energetic
particles.
- Radiocarbon Carbon-14, which is used to date dead plant and animal
matter. Radiocarbon is not used for dating rocks.
- Radiometric dating Determination of a time interval (e.g., the time
since formation of a rock) by means of the radioactive decay of its material.
Radiometric dating is one subset of the many dating methods used in geology.
- Three-isotope plot In dating, this is a plot in which one axis
represents the parent isotope and the other axis represents the daughter
isotope. Both parent and daughter isotopes are ratioed to a daughter element
isotope that is not produced by radioactive decay. This type of plot gives the
age independent of the original amounts of the isotopes.
- Tree ring A ring visible in the stump or sawed section of a tree
which indicates how much it grew in a year. The age of a tree can be
determined by counting the growth rings.
- Two-component mixing The mixing of two different source materials
to produce a rock. On rare occasions this can result in an incorrect age for
certain techniques that use three-isotope plots. Two component mixing can be
recognized if more than one dating technique is used, or if surrounding rocks
are dated.
- Xenolith Literally, a foreign chunk of rock within a rock. Some
rocks contain pieces of older rocks within them. These pieces were ripped off
the magma chamber in which the main rock formed and were incorporated into the
rock without melting. Xenoliths do not occur in most rocks, and they are
usually recognizable by eye where they do occur. If unrecognized, they can
result in an incorrect date for a rock (the date may be of the older
xenolith).
FOOTNOTES
1 In only a couple of special cases have any decay
rates been observed to vary, and none of these special cases apply to the dating
techniques discussed here. These exceptions are discussed later.
2 The term isotope subdivides elements into groups of atoms that
have the same atomic weight. For example carbon has isotopes of weight 12, 13,
and 14 times the mass of a nucleon, referred to as carbon-12, carbon-13, or
carbon-14 (abbreviated as 12C, 13C, 14C). It is only the carbon-14 isotope that
is radioactive.
breitling,rolex day date,
fake rolex for sale,patek philippe,rado,u boat,
fake rolex for sale,rolex prince,bell ross,corum,rolex daytona,iwc,
replica tag heuer,cartier,hublot,roger dubuis,rolex submariner,
faker rolex for sale,tissot